Coordination CompoundHard
Question
Of the following complex ions, which is diamagnetic in nature ?
Options
A.[ Ni ( CN)4]2-
B.[CuCl4]2-
C.[CoF6]3-
D.[NiCl4]2-
Solution
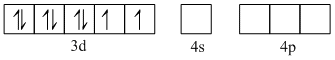
Electronic configuration of Ni2+ in [NiCl4]2- is
Ni2+ = 3d8 4s0
Ni2+ has dsp2 hybridisation, as CN2+ is a strong field ligend.
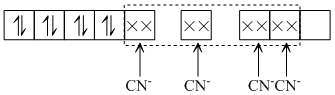
∴ [ Ni ( CN)4]2- is diamagnetic (because of the absence of unpaired electrons).
Ni2+ = 3d8 4s0

Ni2+ has dsp2 hybridisation, as CN2+ is a strong field ligend.

∴ [ Ni ( CN)4]2- is diamagnetic (because of the absence of unpaired electrons).
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Crystal field stabilization energy for high spin d4 octahedral complex is...Na2S + Na2[Fe(CN)5NO] → Na4[Fe(CN)5NOS] ; oxidation number of Fe in reactant (complex) and product (complex) are :...Gun metal is:...NiCl2{P(C2H5)2(C6H5)}2 exhibits temperature dependent magnetic behaviour (paramagnetic/diamagnetic).The coordination geo...The diamagnetic species is :...