Coordination CompoundHard
Question
NiCl2{P(C2H5)2(C6H5)}2 exhibits temperature dependent magnetic behaviour (paramagnetic/diamagnetic).The coordination geometries of Ni2+ in the paramagnetic and diamagnetic states are respectively
Options
A.tetrahedral and tetrahedral
B.square planar and square planar
C.tetrahedral and square planar
D.square planar and tetrahedral
Solution
In both complexes Ni exists as Ni2+.
In sp3 (tetrahedral)
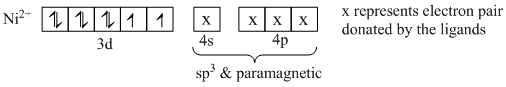
In dsp2 (square planar geometry)
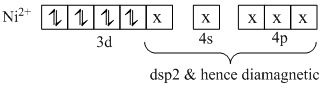
In sp3 (tetrahedral)

In dsp2 (square planar geometry)
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The number of sigma bonds in Zeise′s salt is:...The organometallic compound is :...Silver cholride dissolves in :...Among the following, metal carbonyls, the C − O bond is strongest :...The population p(t) at time t of a certain mousespecies satisfies the differential equation = 0.5 p(t) - 450. If p(0) = ...