Coordination CompoundHard
Question
Crystal field stabilization energy for high spin d4 octahedral complex is
Options
A.- 1.8 ᐃo
B.- 1.6 ᐃo + P
C.- 1.2 ᐃo
D.- 0.6 ᐃo
Solution
In case of high spin complex, ᐃo is small. Thus, the energy required to pair up the fourth electron with the electrons of lower energy d-orbitals would be higher than that required to place the electrons in the higher d-orbital. Thus, pairing does not occur.
For high spin d octahedral complex,
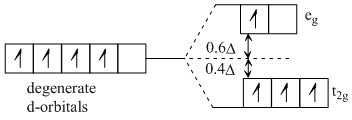
∴ Crystal field stabilisation energy
= (- 3 × 0.4 + 1 × 0.6) ᐃo
= (- 1.2 + 0.6) ᐃo
= - 0.6 ᐃo
For high spin d octahedral complex,

∴ Crystal field stabilisation energy
= (- 3 × 0.4 + 1 × 0.6) ᐃo
= (- 1.2 + 0.6) ᐃo
= - 0.6 ᐃo
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Philosopher′s wool on heating with BaO at 1100oC produces:...The colourless species is :...Which of the following complex does not show geometrical isomerism ?...On adding AgNO3 solution to a solution of [Pt(NH3)3Cl3]Cl, the percentage of total chloride ion precipitated is:...Which one of the following statement is false for nickel-dimethylglyoximate complex ?...