Chemical Kinetics and Nuclear ChemistryHard
Question
A reaction is catalysed by H+ ion;and in the rate law the dependence of rate is of first order with respect to the concentration of H+ ions, in presence of HA rate constant is 2 × 10-3 min-1 and in presence of HB rate constant is 1 × 10-3 min-1. HA and HB have relative strength as :
Options
A.0.5
B.0.002
C.0.001
D.2
Solution
We know
Rate = k [conc.]
Given Rxn catalysed by HA and HB
Rate constant kA = k1 [H+]A
kB = k1 [H+]B
Then relative strength of acids A and B is
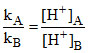
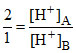 = strength of
= strength of 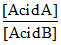
Rate = k [conc.]
Given Rxn catalysed by HA and HB
Rate constant kA = k1 [H+]A
kB = k1 [H+]B
Then relative strength of acids A and B is
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
First order reaction 2N2O(g) → 2N2g + O2(g)has a rate constant of 1.3 × 10-11 S-1 at 270C and 4.5 × 10-1...For a first order reaction, (A) → products, theconcentration of A changes from 0.1 M to0.025 M in 40 minutes. The ...Which of the following statements are true for chemisorption...2713Al is a stable element. 2913Al is expected to disintegrate by :...The colligative property is not represented by :...