Chemical BondingHard
Question
If
(i) C + O2 → CO2
(ii) C +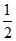 O2 → CO
O2 → CO
(iii) CO +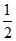 O2 → CO2
O2 → CO2
The heats change for (i), (ii), and (iii) reactions are Q, -12, -10 respectively. Then Q = ?
(i) C + O2 → CO2
(ii) C +
(iii) CO +
The heats change for (i), (ii), and (iii) reactions are Q, -12, -10 respectively. Then Q = ?
Options
A.-2
B.-22
C.2
D.-16
More Chemical Bonding Questions
After understanding the assertion and reason, choose the correct option.Assertion : In the bonding molecular orbital (MO...For the following reaction if equal mass of A and B are taken : A + 2B → CWhich of the following is/are correct ? ...Which of the following molecule has highest dipole moment ?...Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectivel...The critical temperature of water is higher than that of O2 because the H2O molecule has :...