Chemical BondingHard
Question
Which of the following molecule has highest dipole moment ?
Options
A.B2H6
B.NF3
C.NH3
D.BF3
Solution
BF3 and B2H6 being none-polar molecules do not show dipole moment. in NF3, The electronegative F pulls the electron toward itself due to which bond moments of the three N - F bonds is opposite to that of lone pair while in NH3 bond moments of the N - H bonds are in same are in same direction to that of lone pair.
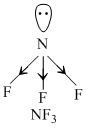
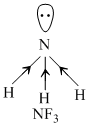
Hence, bond moments add up in NH3 resulting in highest dipole moment among the given options.


Hence, bond moments add up in NH3 resulting in highest dipole moment among the given options.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Incorrect order of bond angle is:-...Which one of the following pairs of species have the same bond order?...N2 and O2 are converted into monocations, N2+ and O2+ respectively.Which of the following is worng...NaCl, NaBr, NaI mixture on adding H2SO4 gives gases respectively :-...During change of O2 to O2- ion, the electron adds on which one of the following orbitals?...