JEE Advanced | 2015Chemical BondingHard
Question
Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectively, are
Options
A.CH3SiCl3 and Si(CH3)4
B.(CH3)2SiCl2 and (CH3)3SiCl
C.(CH3)2SiCl2 and CH3SiCl3
D.SiCl4 and (CH3)3SiCl
Solution
(A) CH3 SiCl3 
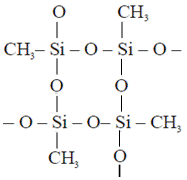 Cross linked polymer
Cross linked polymer
Si(CH3)4 → NOT hydrolysed
(B) (CH3)2 SiCl2
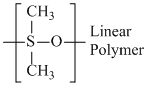
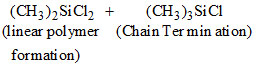
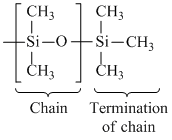
(C) (CH3)2SiCl2 linear polymer formation
linear polymer formation
CH3SiCl3 Cross linked polymer
Cross linked polymer
(D) SiCl4 H4SiO4 + 4HCl
H4SiO4 + 4HCl
 Cross linked polymer
Cross linked polymerSi(CH3)4 → NOT hydrolysed
(B) (CH3)2 SiCl2
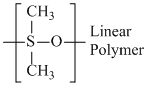

(C) (CH3)2SiCl2
CH3SiCl3
(D) SiCl4
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
A + H2O → B + HClB + H2O → HClCompound (A) , (B) and (C) will be respectively :-...Which one of the following arrangements of molecules is correct on the basis of their dipole moments?...Consider the following species: CN+, CN–, NO and CN Which one of these will have the highest bond order?...If(i) C + O2 → CO2(ii) C + O2 → CO(iii) CO + O2 → CO2The heats change for (i), (ii), and (iii) reactio...Which one among the following does not have the hydrogen bond ?...