Chemical BondingHard
Question
For the following reaction if equal mass of A and B are taken :
A + 2B → C
Which of the following is/are correct ? (MA and MB are molar masses of A and B respectively)
A + 2B → C
Which of the following is/are correct ? (MA and MB are molar masses of A and B respectively)
Options
A.If MA = 2MB, then none of the reactant will be left
B.If MB > 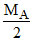 , then A will be limiting reagent
, then A will be limiting reagent
C.If MA = MB, then A will be limiting reagent
D.MB > MA the B will be limiting reagent
More Chemical Bonding Questions
Assuming 2s-2p mixing is NOT operative, the paramagnetic species among the following is...Amongst LiCl, RbCl BeCl2 and MgCl2, the compounds with the greatest and the least jonic character, respectively are :-...Which of the following arrangements does not represent the correct order of the property stated against it ?...The correct stability order for the following species is :-...For the rx :- CO(g) + O2(g) ⇋ CO2(g) is :...