Gaseous StateHard
Question
The average kinetic energy of an ideal gas per molecule in S.I. units at 25oC will be ?
Options
A.6.17 × 10-21 KJ
B.6.17 × 10-21 J
C.6.17 × 10-20 J
D.6.17 × 16-20 J
Solution
Avg. K.E. = - 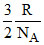
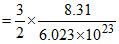 × 298
× 298
= 6.17 × 10-21 J
= 6.17 × 10-21 J
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
In the figure-2 the pressure of the confined gas will be...The curve of pressure volume (PV) against pressure (P) of the gas at a particular temperature is as shown, according to ...A reversible cyclic process for an ideal gas is shown below. Here, P , V and T are pressure , Volume and temperature , r...According to kinetic theory of gases, for a diatomic molecule :...The volume of a gas increases by a factor of 2 while the pressure decreases by a factor of 3. Given that the number of m...