Atomic StructureHard
Question
The neon atoms has a radius of 160 pm. What is the edge of the unit cell of a face centered structure of neon ?
Options
A.490 pm
B.320 pm
C.453 pm
D.481 pm
Solution
r = 160 pm
for fcc 4r = a√2 a =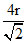
a =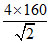 = 453 pm
= 453 pm
for fcc 4r = a√2 a =
a =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If the value of En = - 78.4 kcal/mole, the order of the orbit in hydrogen atom is :...Five valence electrons of nitrogen are shown as : If the spin quantum number of B and E is -1/2. The group of electrons ...Rutherford′s α-particle scattering experiment eventually led to thje conclusion that :...Energy of an electron is given by E = - 2.178 × 10-18 J Wavelength of light required to excite an electron in an hy...Which of the following sets of quantum numbers represents the highest energy of an atom?...