Atomic StructureHard
Question
Five valence electrons of nitrogen are shown as :
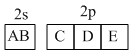
If the spin quantum number of B and E is -1/2. The group of electrons with three same quantum numbers are :-

If the spin quantum number of B and E is -1/2. The group of electrons with three same quantum numbers are :-
Options
A.Only AB
B.Only CDE
C.Both the above
D.None of these
More Atomic Structure Questions
A variable, opposite external potential (Eext) is applied to the cell Zn|Zn2+ (1 M) || Cu2+ (1M) | Cu, of potential 1.1 ...In a sample of H-atoms, electrons de-excite from a level ′n′ to 1. The total number of lines belonging to Ba...Calculate the energy in joule corresponding to light of wavelength 45 nm : (Planck′s constant h = 6.63 × 10-3...Correct Match is :-...The magnetic moment of Nix+ ion (Z = 28) is about 2.82 BM. The value of x is...