ThermodynamicsHard
Question
Two adiabatic processes involving an ideal gas are plotted on a P - V diagram. A and B are two points on these curves as shown in the diagram. Which of the following statements is TRUE?
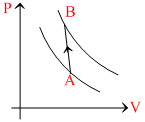

Options
A.Heat is given to system in the process AB
B.Heat is rejected by system in the process AB
C.Whether heat is rejected or absorbed by the system during the process AB depends on the magnitude of the slope of line AB
D.Whether heat is rejected or absorbed by the system during the process AB depends on the position of the points A and B on the P - V diagram.
Solution
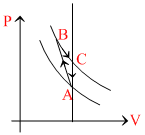
ᐃVABCA = 0, wABCA = + z
For process B to C
QB → C = 0
WB → C = x, ᐃVB → C = - x
for process (Isochoric) C to A
⇒ ᐃVC → A = - y ⇒ QC → A = - y
QABCA = WABCA
⇒ QA → B + QB → C + QC → A = + z
⇒ QA → B + 0 + C - y) = + z
⇒ QA → B = z + y > 0
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In the above question, the curve for which the heat is absorbed from the surroundings is :-...A quantity of air (γ = 1.4) at 27oC is compressed suddenly, the temperature of the air system will :...The average kinetic energy of a gas molecule at 27oC is 6.21 × 10-21J, then its average kinetc energy at 227oC is :...If the coefficient of Linear expansion of a solid is 0.00009. Its coefficient of volume expansion is :...Two identical containers A and B with friction pistons contain the ideal gas at the same temperature and the same volume...