ThermodynamicsHard
Question
The average kinetic energy of a gas molecule at 27oC is 6.21 × 10-21J, then its average kinetc energy at 227oC is :
Options
A.10.35 × 10-21 J
B.11.35 × 10-21 J
C.52.2 × 10-21 J
D.5.22 × 10-21 J
Solution
Average kinetic energy of gas molecules ∝ Temperature (Absolute)
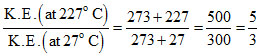
K.E.(227o) =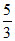 × 6.21 × 10-21 J
× 6.21 × 10-21 J
= 10.35 × 10-21
K.E.(227o) =
= 10.35 × 10-21
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Fraunh offer line the solar system is an example of :...Two metallic spheres S1 and S2 are made of the same material and have got identical surface finish. The mass of S1 is th...An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 a...An ideal gas expands in such a way that PV2 = constant throughout the process. Select correct alternative...For an adiabatic expansion of a perfect gas, the value of DP/P is equal to:-...