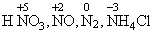Redox and Equivalent ConceptHard
Question
Which ordering of compounds is according to the decreasing order of the oxidation state of nitrogen?
Options
A.HNO3, NO, NH4Cl, N2
B.HNO3, NO, N2, NH4Cl
C.HNO3, NH4Cl, NO, N2
D.NO, HNO3, NH4Cl, N2
More Redox and Equivalent Concept Questions
Which of the following ion cannot act as an oxidizing agent?...In which of the following compounds, nitrogen exhibits highest oxidation state?...In which of the following reactions H2O2 acts as a reducing agent? (a) H2O2 + 2H+ + 2e- → 2H2O (b) H2O2 - 2e- U...An aqueous solution of 6.3 g oxalic acid dihydrate is made up to 250 mL. The volume of 0.1 N NaOH required to completely...The oxidation state of chromium in the final product formed by the reaction between KI and acidified potassium dichromat...