Solid StateHard
Question
A compound MpXq has cubic close packing (ccp) arrangement of X. Its unit cell structure is shown below. The empirical formula of the compound is
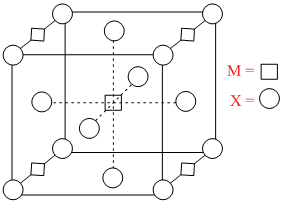

Options
A.MX
B.MX2
C.M2X
D.M5X14
Solution
X = 8 × 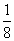 + 6 ×
+ 6 × 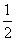 = 4
= 4
M = 4 ×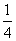 + 1 = 2
+ 1 = 2
So, unit cell formula of the compound is M2X4 and the empirical formula of the compound is MX2.
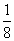 + 6 ×
+ 6 × 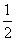 = 4
= 4 M = 4 ×
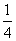 + 1 = 2
+ 1 = 2So, unit cell formula of the compound is M2X4 and the empirical formula of the compound is MX2.
Create a free account to view solution
View Solution FreeMore Solid State Questions
In a CCP lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers.Then the formula of th...The ionic radii of Cs and Cl are 0.165 nm and 0.181nm respectively. Their atomic weights are 133 and 35.5. Then,...Which of the following oxide shows electrical properties like metals?...A mineral having the formula AB2 crystallizes in the CCP lattice with the A atoms occupying the lattice points. What is ...The compound AB crystallizes in a cubic lattice in which both A and B atoms have coordination numbers of 8. To what crys...