Solid StateHard
Question
In a CCP lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers.Then the formula of the compound would be if one of the X atoms from a corner is replaced by Z atoms (also monovalent)?
Options
A.X7Y24Z2
B.X7Y24Z
C.X24Y7Z
D.XY24Z
Solution
X = 7 × 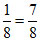 ; Y =
; Y = 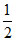 × 6 = 3. ; z =
× 6 = 3. ; z = 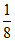 ⇒ X7/8 Y3 Z1/8 = X7 Y24 Z
⇒ X7/8 Y3 Z1/8 = X7 Y24 Z
Create a free account to view solution
View Solution FreeMore Solid State Questions
Frenkel defect is observed in...Number of 3rd nearest neighbour atom in a FCC unit cell are -...In Na2O structure,...Square packed sheets are arranged on the top of other such that a sphere in the next layer rests on the center of a squa...A transition metal M can exist in two oxidation states +2 and +3. It forms an oxide whose experimental formula is given ...