Solid StateHard
Question
Diamond structure can be considered as ZnS (Zinc blend) structure in which Zn2+ in alternate tetrahedral void and S2- in cubic close pack arrangement is replaced by carbon atoms. If C - C covalent bond length in diamond is 1.5 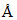 , what is the edge length of diamond unit cell (z = 8) :
, what is the edge length of diamond unit cell (z = 8) :
Options
A.3.46 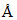
B.6.92 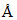
C.1.73 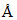
D.3 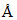
Solution
4x = √3a ⇒ = 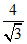 x
x
⇒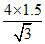 = 2 ×
= 2 × 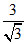 = 2 × √3
= 2 × √3 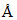 = 1.73 × = 3.46
= 1.73 × = 3.46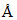
⇒
Create a free account to view solution
View Solution FreeMore Solid State Questions
Three lines are drawn from a single corner of an FCC unit cell to meet the other corner such that they are found to pass...There are three cubic unit cells A, B and C. A is FCC and all of its tetrahedral voids are also occupied. B is also FCC ...Consider a cube 1 of Body Centered Cubic unit cell of edge length a now atom at the body center can be viewed to be lyin...The correct statement(s) regarding defects in solids is(are)...In a CCP lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers.Then the formula of th...