Solid StateHard
Question
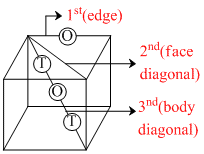
Three lines are drawn from a single corner of an FCC unit cell to meet the other corner such that they are found to pass through exactly only 1 octahedral void, no voids of any type and exactly 2 tetrahedral voids with 1 octahedral void. Identify the line in the same order.
Options
A.Edge, Face diagonal, Body diagonal
B.Face diagonal, Edge, Body diagonal
C.Body diagonal, Face diagonal, Edge
D.Edge, Body diagonal, Face diagonal
More Solid State Questions
What type of crystal defect is indicated in the diagram below ?Na+ Cl- Na+ Cl- Na+ Cl-Cl- - Cl- Na+ - Na+Na+ Cl- - ...In each of the following ionic solids, the coordination number of the cation and anion is same, except...Chromium metal crystallises in body centred cubic lattice. The edge length of unit cell is found to be 287 pm. Atomic ra...BaO has a rock salt type structure. When subjected to high pressure, the ratio of the coordination number of Ba2+ ion to...An ionic compound consists of only A+, B2+ and C3− ions. Which of the following option(s) is/are correct with respect to...