ThermodynamicsHard
Question
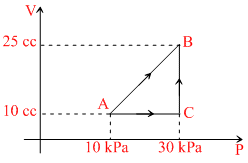
Figure shows two paths through a gas can be taken from the state A to the state B. The work done by the gas which is higher than the other is :-

Options
A.1.5 J
B.2 J
C.0.45 J
D.0.6 J
Solution
ᐃWAB = Area under A → B bounded by volume axis.
= 10 × 103 (25 - 10) × 10-6 +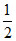 (25 - 10) × 10-6 × 20 × 103 = 0.15 + 0.15 = 0.3 J.
(25 - 10) × 10-6 × 20 × 103 = 0.15 + 0.15 = 0.3 J.
In path A → C → B = 30 × 103 (25 - 10) × 10-6 = 0.45 J.
= 10 × 103 (25 - 10) × 10-6 +
In path A → C → B = 30 × 103 (25 - 10) × 10-6 = 0.45 J.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
When a gas is adiabatically compressed then it′s temp. increase because : -...The curves shown represent adiabatic curves for monoatomic, diatomic & polyatomic (γ = 4/3) gases. The slopes for c...If an ideal gas compressed during isothermal process then :-If an ideal gas compressed during isothermal process then :-...To rigid boxes containing different ideal gases are placed on a table. Box A contains one mole are placed on a table. Bo...An ideal gas is taken around the cycle ABCA as shown in the P - V diagram. The net work done by the gas during the cycle...