ThermodynamicsHard
Question
The curves shown represent adiabatic curves for monoatomic, diatomic & polyatomic (γ = 4/3) gases. The slopes for curves 1,2,3 respectively at point A are-
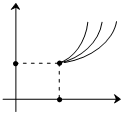

Options
A.2.5 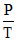 , 3.5
, 3.5 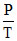 , 4.5
, 4.5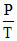
B.2.5 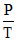 , 3
, 3 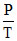 , 4
, 4 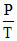
C.2.5 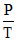 , 3.5
, 3.5 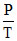 , 4
, 4 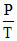
D.4 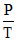 , 3.5
, 3.5 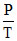 , 2.5
, 2.5 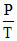
Solution
For adiabatic process : Slope : 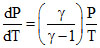
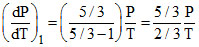 = 2.5
= 2.5 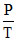
 = 3.5
= 3.5 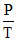
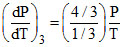 = 4
= 4 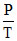
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
If the heat of 110 J is added to a gaseous system, change in internal energy is 40 J, then the amount of external work d...An ideal gas is taken around the cycle ABCA as shown in the P - V diagram. The net work done by the gas during the cycle...The following graphs between P and V are plotted at two temperature T1 and T2. Then which of the following is correct?...The imstryment used to measure the temperature of the source from its thermal radiation is :...Steam at 100o C is passes into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15oC till the t...