Solid StateHard
Question
Which of the following atoms touch each other in a fcc unit cell (diagram given) -
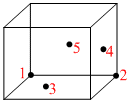
Atom 4 is at the centre of side face
Atom 5 is at the centre of back face
Atom 3 is at the centre of bottom face

Atom 4 is at the centre of side face
Atom 5 is at the centre of back face
Atom 3 is at the centre of bottom face
Options
A.1 & 2
B.1 & 3
C.3 & 5
D.3 & 4
More Solid State Questions
A mineral having the formula AB2 crystallizes in the CCP lattice with the A atoms occupying the lattice points. What is ...If the height of HCP unit cell of identical particles is h, then the height of tetrahedral voids from the base is...MgO exist in a rock-salt type unit cell. Each Mg+2 ion will be in contact with...A metallic element exists as cubic lattice. Each edge of the unit cell is 4.0 Å. The density of the metal is 6.25 g/cm3....A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal ...