Solid StateHard
Question
A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is
Options
A.288 pm
B.408 pm
C.144 pm
D.204 pm
Solution
For fcc lattice,
4r = √2a
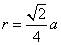
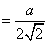
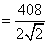
= 144 pm;
diameter d = 2r = 2 × 144 pm
= 288 pm
4r = √2a
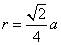
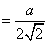
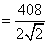
= 144 pm;
diameter d = 2r = 2 × 144 pm
= 288 pm
Create a free account to view solution
View Solution FreeMore Solid State Questions
A mineral having the formula AB2 crystallizes in the CCP lattice with the A atoms occupying the lattice points. What is ...If the unit cell length of sodium chloride crystal is 600 pm, then its density will be...In Na2O structure,...In a face centered cubic cell, the contribution of an atom at a face of the unit cell is :...An alkali metal has density 4.5 g/cm3. It has cubic unit cell with edge length 400 pm. The reaction of 7.68 cm3 chunk of...