Chemical EquilibriumHard
Question
For the given reaction at constant pressure,
n A (g) ⇋ An (g)
Initial moles 1 0
Moles at equilibrium 1 - α α/n
Then the correct relation between initial density (di) & final density (df) of the system is
n A (g) ⇋ An (g)
Initial moles 1 0
Moles at equilibrium 1 - α α/n
Then the correct relation between initial density (di) & final density (df) of the system is
Options
A.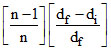 = α
= α
B.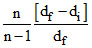 = α
= α
C.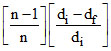 = α
= α
D.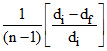 = α
= α
Solution
Total moles at equilibrium = 1 - α + α/n = 1 + 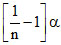
So using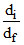 = 1 +
= 1 + 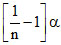
So using
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
When CO2(g) is dissolved in water, then the following equilibrium is establishedCO2(aq) + 2H2O(l) $\rightleftharpoons$H3...For the reaction H2(g) + I2(g) ⇋ 2HI(g) Kc = 66.9 at 350oC and Kc = 50.0 at 448oC. The reaction has...At 525 K, PCl5(g) is 80% dissociated at a pressure of 1 atm. Now, sufficient quantity of an inert gas at constant pressu...In the reaction C(s) + CO2(g) ⇋ 2CO(g), the equilibrium pressure is 12 atm. If 50% of CO2 reacts then Kp will be :...In a closed rigid vessel, the following equilibrium partial pressures are measured, where N2 = 100 mm, H2 = 400 mm and N...