Chemical EquilibriumHard
Question
The equilibrium constant for the reaction
SO3(g) ⇋ SO2(g) +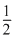 O2(g)
O2(g)
is Kc = 4.9 × 10-2. The value of Kc for the reaction
2SO2(g) + O2(g) ⇋ 2SO3(g)
will be
SO3(g) ⇋ SO2(g) +
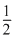 O2(g)
O2(g)is Kc = 4.9 × 10-2. The value of Kc for the reaction
2SO2(g) + O2(g) ⇋ 2SO3(g)
will be
Options
A.416
B.2.40 × 10-3
C.9.8 × 10-2
D.4.9 × 10-2
More Chemical Equilibrium Questions
For a given exothermic reaction, Kp and K′p are the equilibrium constants at temperatures T1 and T2, respectively....One mole each of A and B and 3 moles each of C and D are placed in 1 L flask. If equilibrium constant is 2.25 for the re...At 525 K, PCl5(g) is 80% dissociated at a pressure of 1 atm. Now, sufficient quantity of an inert gas at constant pressu...An amount of 3 moles of N2 and some H2 is introduced into an evacuated vessel. The reaction starts at t = 0 and equilibr...Which of the following statements is correct for a reversible process in a state of equilibrium ?...