Atomic StructureHard
Question
An electrons of a stationary hydrogen atom passes from the fifth energy level to the ground level. Thevelocity that the atom acquired as a result of photon emission will be
(m is the mass of the electron, R, Rydberg constant and h, Planck′s constant)
(m is the mass of the electron, R, Rydberg constant and h, Planck′s constant)
Options
A.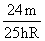
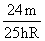
B.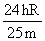
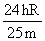
C.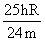
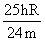
D.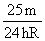
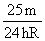
Solution
Momentum of atom, P = 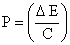
Velocity of H2 atom =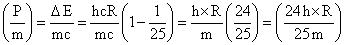
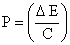
Velocity of H2 atom =
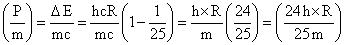
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The angular momentum of an electron in the 2nd excited state of a Helium ion (He+) is...X-ray will not show the phenomenon of :...In 88Ra226 nucleus there are :...Hydrogen (1H1), Deuterium (1H2) singly ionised Helium (2He4)+ and doubly ionised lithium (3Li6)++; all have one electron...In nucleus of mass number A, originally at rest, emits an a - particle with speed v. The daughter nucleus recoils with a...