Atomic StructureHard
Question
The angular momentum of an electron in the 2nd excited state of a Helium ion (He+) is
Options
A.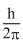
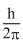
B.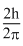
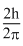
C.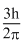
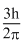
D.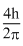
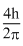
Solution
Angular momentum in the ground state = 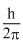
Angular momentum in the first excited state =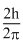
Angular momentum in the second exited state =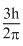
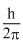
Angular momentum in the first excited state =
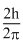
Angular momentum in the second exited state =
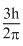
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Photoelectric effect supports quantum nature of light because...If an alpha particle with energy 7.7 MeV is bombarded on a thin gold foil, the closest distance from nucleus it can reac...Which statement is correct :-...For the same rise in temperature of one mole of gas at constant volume the heat required for non-linear triatomic gas is...Mark the INCORRECT statement :-...