Coordination CompoundHard
Question
Wilkinson′s catalyst react with H2 to form an octahedral complex in which Rh (Z = 45) has the following electronic configuration in the ligand field, t2g2,2,2, eg0,0
Then which of the following is correct about this new complex ?
Then which of the following is correct about this new complex ?
Options
A.It is diamagnetic
B.Its IUPAC name is chloridotris(triphenylphosphine)rhodium(I)
C.Hybridisation of Rh (I) is sp3d2
D.It is a paramagnetic complex
Solution
All electrons are paired ; so diamagnetic and has d2sp3 hybridisation.
[RhCl(PPh3)3] + H2 [RhCl(H)2(PPh3)3]
Wilkinsons′s catalyst
New complex chloridodihydridotris(triphenyl phosphine)rhodium(III)
Rh(III) → 4d6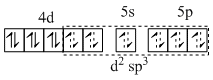
[RhCl(PPh3)3] + H2 [RhCl(H)2(PPh3)3]
Wilkinsons′s catalyst
New complex chloridodihydridotris(triphenyl phosphine)rhodium(III)
Rh(III) → 4d6

Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
[Co(NH3)5NO2]Cl2 and [Co(NH3)5ONO]Cl2 are related to each other as :...50 ml of 0.2 M solution of a compound with empirical formula CoCl3.4NH3 on treatment with excess of AgNO3(aq) yields 1.4...Gun metal is:...The crystal field-splitting for Cr3+ ion in octahedral field changes for ligands I-, H2O, NH3, CN- and the increasing or...Which of the following statements is false ?...