Coordination CompoundHard
Question
The compound Na2IrCl6 reacts with triphenylphosphine in diethyleneglycol in an atmosphere of CO to give [IrCl(CO)(PPh3)2]. (Atomic number of Ir = 77)
Which of the following statements is /are correct?
Which of the following statements is /are correct?
Options
A.The IUPAC name of the complex is carbonylchloridobis(triphenylphosphine)iridium(I).
B.The hybridisation of the metal ion is sp3
C.The magnetic moment (spin only) of the complex is zero
D.The complex shows geometrical as well as ionization isomerism
Solution
(A) [IrCl(CO)(PPh3)2] carbonylchloridobis(triphenylphosphine)iridium(I).
(B) Coordination number of Ir is four. Ir is in (+1) oxidation state with 4d8 configuration. It is trans isomer, so its geometry should be square planar.
[IrCl(CO)(PPh3)2]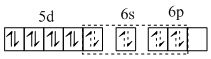
(C) All electrons are paired ; so magnetic moment is zero.
(D)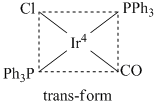
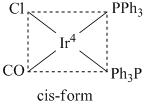
The complex has plane of symmetry, so it does not show optical isomerism.
(B) Coordination number of Ir is four. Ir is in (+1) oxidation state with 4d8 configuration. It is trans isomer, so its geometry should be square planar.
[IrCl(CO)(PPh3)2]

(C) All electrons are paired ; so magnetic moment is zero.
(D)


The complex has plane of symmetry, so it does not show optical isomerism.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which one of the following statements concerning lanthanide elements is false ?...The magnitude of crystal field stabilisation energy in octahedral field depends on I: the nature of the ligand II: the c...The EAN of metal atoms in [Fe(CO)2(NO+)2] and Co2(CO)8 respectively are :...Which of the following does not have optical isomer ?...The [Fe(CN)6]3- complex ion...