Coordination CompoundHard
Question
In which of the following pairs both the copmplexes show optical isomerism?
Options
A.cis - [Cr(C2O4)2Cl2]3-, cis - [co(NH3)4Cl2]
B.[CO(en)3]Cl3, cis - [Co(en)2Cl2]Cl
C.[PtCl(dien)]Cl, [NiCl2Br2]2-
D.[Co(NO3)2(NH3)3], cis - [Pt(en)2Cl2]
Solution
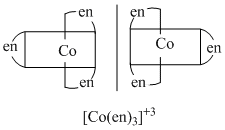
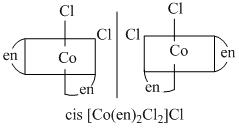
They form non-superimposable mirror image, so they are optically active compounds.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following statements is not correct?...Trioxalato aluminate (III) and tetrafluoro-borate (III) ions are respectively...A complex of certain metal has the magnetic moment of 4.91 BM whereas another complex of the same metal with same oxidat...Nickel (Z = 28) combines with a uninegative monodentate ligand X- to form a paramagnetic complex [NiX4]2-. The number of...Which of the following complexes show geometrical as well as optical isomerism ?(1) [Cr(OX)3]3-(2) [Rh(en)2Cl2]+(3) [Co(...