p-Block elementsHard
Question
The true statement for the acids of phosphorus. H3PO2, H3PO3, and H3PO4 is :
Options
A.the order of their acidity is H3PO4 < H3PO3 < H3PO2
B.all of them are reducing in nature
C.all of them are tribasic acids
D.the geometry of phoshorus is tetrahedral in all the the three
Solution
(i) The geometry of phosphorus is tetrahedral in all the three.
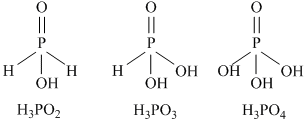
(ii) The acidity increases with increase in oxidation number of central atom
+1 +3 +5
H3PO2 < H3PO3 < H3PO4
(iii) H3PO4 is not reducing.
(iv) H3PO3 is diabasic while H3PO2 is monobasic.

(ii) The acidity increases with increase in oxidation number of central atom
+1 +3 +5
H3PO2 < H3PO3 < H3PO4
(iii) H3PO4 is not reducing.
(iv) H3PO3 is diabasic while H3PO2 is monobasic.
Create a free account to view solution
View Solution Free