p-Block elementsHard
Question
Which of the following statement(s) is/are correct ?
Options
A.The oxide, B2O3 and B(OH)3 are acidic
B.The halides of B (except BF3) and Si are readily hydrolysed.
C.The hydrides of B and Si are volatile, spontaneously flammable and readily hydrolysed.
D.Aluminium hydride is a polymer, (AlH3)n.
Solution
(A) It is a acidic oxide and is anhydride of boric acid and it reacts with alkalies or bases to form borates.
3Na2O + B2O3 → 2Na3BO3 (sodium orthoborate)
It is a weak monobasic acid soluble in water and in aqueous solution the boron atom completes its octet by accepting OH- from water molecules:
B(OH)3(aq) + 2H2O(l) ⇋ [B(OH)4]- (aq) + H3O + (aq). pK = 9.25.
(B) BX3 (except BF3) get hydrolysed due to presence of vacant p-orbital and SiX4 get hydrolysed due to presence of vacant d-orbital.
(C) Controlled pyrolysis of diborane leads to most of the higher boranes.
It catches fire spontaneously in air and explodes with O2. Reaction with oxygen is extremely exothermic.
B2H6 + 3O2 → B2O3 + 3H2O ᐃH = - 2160 kJ mol-1
Si2H1 + (4 + n) H2O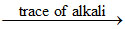 2SiO2 + nH2O + 7H2
2SiO2 + nH2O + 7H2
(D) Aluminium hydride is a polymer due to formation of electron defecient bond (Al − H--- Al).
3Na2O + B2O3 → 2Na3BO3 (sodium orthoborate)
It is a weak monobasic acid soluble in water and in aqueous solution the boron atom completes its octet by accepting OH- from water molecules:
B(OH)3(aq) + 2H2O(l) ⇋ [B(OH)4]- (aq) + H3O + (aq). pK = 9.25.
(B) BX3 (except BF3) get hydrolysed due to presence of vacant p-orbital and SiX4 get hydrolysed due to presence of vacant d-orbital.
(C) Controlled pyrolysis of diborane leads to most of the higher boranes.
It catches fire spontaneously in air and explodes with O2. Reaction with oxygen is extremely exothermic.
B2H6 + 3O2 → B2O3 + 3H2O ᐃH = - 2160 kJ mol-1
Si2H1 + (4 + n) H2O
(D) Aluminium hydride is a polymer due to formation of electron defecient bond (Al − H--- Al).
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following metals does not dissolve in concentrated HNO3?...How many chiral compounds are possible on monochlorination of 2-methyl butane ?...Iodide of Millon′s base is...When orthoboric acid (H3BO3) is heated the residue left is :...Which of the following gives acetylene on hydrolysis :...