p-Block elementsHard
Question
When 8.3 g copper sulphate reacts with excess of potassium iodide then the amount of iodine liberated is :
Options
A.42.3 g
B.24.3 g
C.4.23 g
D.2.43 g
Solution
2CuSO4. 5H2O + 4Kl → Cu2I2 + 2K2SO4 + I2 + 10H2O
498g 254g
498g of CuSO4 liberate I2 = 254g
8.3 g of CuSO4 liberate I2 =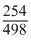 × 8.3
× 8.3
= 4.23 g on treatment with iodine produces :
498g 254g
498g of CuSO4 liberate I2 = 254g
8.3 g of CuSO4 liberate I2 =
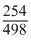 × 8.3
× 8.3= 4.23 g on treatment with iodine produces :
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
In the preparation of HBr or HI , NaX (X = Br, I) is treated with H3PO4 and not by concentrated H2SO4 since,...When zeolite, which is hydrated sodium aluminium silicate, is treated with hard water, the sodium ions are exchanged wit...Among the following molecules, (i) XeO3 (ii) XeOF4 (iii) XeF6 those having same number of lone pairs on Xe are :...In lake test of Al3+ ion, there is formation of coloured floating. It is due to :...Which of the following product is formed by the reaction of sulphur dioxide with chlorine in presence of charcoal ?...