ThermodynamicsHard
Question
A block of ice at -10oC is slowly heated and converted to steam at 100oC. Which of the following curves represents the phenomena qualitative ?
Options
A.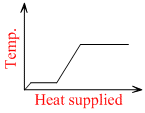

B.

C.

D.

Solution
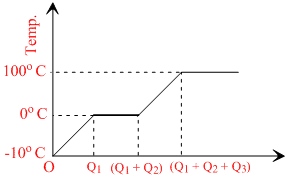
The temperature of ice will first increase from -10o C to 0o C.
Heat supplied in this process will be
Q1 = msi (10)
Where, m = mass of ice
si = specific heat of ice
Then, ice start melting. Temperature during melting will remain constant (0o C).
Heat supplied in this process will be
Q3 = msw(100)
Where, sw = specific heat of water
Finally, water at 100o C will be converted into steam at 100o C and during this process temperature again remains constant. Temperature versus heat supplied graph will be as shown in figure.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
If Cp and Cv denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, ...If an ideal flask containing hot coffee is shaken, the temperature of the coffee will :...Which one of the following is not thermodynamical co-ordinate ?...To rigid boxes containing different ideal gases are placed on a table. Box A contains one mole are placed on a table. Bo...The specific heat of a gas at constant pressure is more than that of the same gas at constant volume because :-...