ThermochemistryHard
Question
Enthalpy of combustion of CH4C2H6 and C3H8 are - 210, - 368.4 and - 526.2 k cal mol-1 respectively. Enthalpy of conbustion of hexane can be predicated as
Options
A.-840k cal mol-1
B.-684k cal mol-1
C.-1000k cal mol-1
D.none of these
Solution
ᐃHc(C2H6) - ᐃHc(CH4)
= -368.2-(-210.8) = - 157.4
ᐃHc(C3H8) - ᐃHc(C2H6)
= - 526.2 - (-368.4) = - 157.8
Thus, average ᐃHc (-CH2) =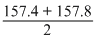
= - 157.6kcal mol-1
Then,
ᐃHc(C6H14) = - ᐃHc(C3H8) + 3ᐃHc(-CH2)
= - 526.2 + 3 (- 157.6) = - 99 k cal
= -368.2-(-210.8) = - 157.4
ᐃHc(C3H8) - ᐃHc(C2H6)
= - 526.2 - (-368.4) = - 157.8
Thus, average ᐃHc (-CH2) =
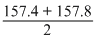
= - 157.6kcal mol-1
Then,
ᐃHc(C6H14) = - ᐃHc(C3H8) + 3ᐃHc(-CH2)
= - 526.2 + 3 (- 157.6) = - 99 k cal
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
When 1.0 g of oxalic acid (H2C2O4) is burned in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature inc...Among the following, for which reaction the heat of reaction represents bond energy of HCl?...Consider the following data:$\Delta_{f}H^{\Theta}($ methane, g$) = - XkJ{mol}^{- 1}$Enthalpy of sublimation of graphite ...For the reaction, 2NO(g) + O2(g) → 2NO2(g); ΔH = −35.0 kcal at 300 K. If 6.0 moles of NO reacts with 3.0 moles of O2 at ...The standard enthalpy combustion at 25oC of hydrogen, cyclohexene (C6H10) and cyclohexane (C6H12) are - 241, - 3800 and ...