ThermochemistryHard
Question
The standard enthalpy combustion at 25oC of hydrogen, cyclohexene (C6H10) and cyclohexane (C6H12) are - 241, - 3800 and - 3920 kj/ mole respectively. Calculate the heat of hydrogenation of cyclothexene.
Options
A.- 111kJ/mole
B.- 121kJ/mole
C.- 118kJ/mole
D.- 128 kJ/mole
Solution
The required reaction is
C6H10 + H12 → C6H12, ᐃH1 = ? .....(i)
Cyclohexeme Cyclohexame
Let us write the give facts
H2 +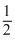 O2
O2 → H2O;
ᐃH2 = - 241 kJ/mole .....(ii)
C6H10 +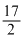 O2
O2 → 6CO2 + 5H2O
H3 = - 3800kJ/mole .....(iii)
C6H12 + 9O2 → 6CO2 + 6H2O
H4 = - 3920kJ/mole ..........(4)
The required reaction rectional (1)Can be obained by adding equations (2) and (3) andsubracting (4) From the sum of (2) and (3).
C6H10 + H29O2 → C6H12
ᐃH1 = (ᐃH2 = ᐃH3) - ᐃH4
= [-241+ (-3800)] - (-3920)
=(-241 - 3800) - (- 3920)
= -4041 + 3920 = - 121kJ/mole
C6H10 + H12
Cyclohexeme Cyclohexame
Let us write the give facts
H2 +
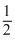 O2
O2 ᐃH2 = - 241 kJ/mole .....(ii)
C6H10 +
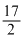 O2
O2 H3 = - 3800kJ/mole .....(iii)
C6H12 + 9O2
H4 = - 3920kJ/mole ..........(4)
The required reaction rectional (1)Can be obained by adding equations (2) and (3) andsubracting (4) From the sum of (2) and (3).
C6H10 + H29O2
ᐃH1 = (ᐃH2 = ᐃH3) - ᐃH4
= [-241+ (-3800)] - (-3920)
=(-241 - 3800) - (- 3920)
= -4041 + 3920 = - 121kJ/mole
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Enthalpies of solution of BaCl2(s) and BaCl2·2H2O(s) are −20.6 kJ/mol and 8.8 kJ/mol, respectively. ΔH hydration of BaCl...If x1, x2 and x3 are enthalpies of H - H, O = O and O - H bonds respectively, and x4 is the enthalpy of vaporisation of ...The specific heats of iodine vapours and solid are 0.031 and 0.055 cal/g, respectively. If the enthalpy of sublimation o...Study the following thermochemical data.S + O2 → SO2: ΔH = −298.2 kJSO2 + ½ O2 → SO3: ΔH = −98.2 kJSO3 + H2O → H2SO4: ΔH...Among the following the reaction for which ΔH = ΔE, is...