ThermochemistryHard
Question
If x1, x2 and x3 are enthalpies of H - H, O = O and O - H bonds respectively, and x4 is the enthalpy of vaporisation of water, estimate the standard enthalpy of combustion of hydrogen.
Options
A.x1 + 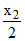 - 2x3 + x4
- 2x3 + x4
B.x1 + 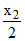 - 2x3 - x4
- 2x3 - x4
C.x1 + 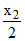 - x3 + x4
- x3 + x4
D.2x3 - x1 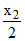 - x4
- x4
Solution
H2 (g) + 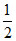 O2 (g) → H2O (l) ; ᐃHcomb,H2
O2 (g) → H2O (l) ; ᐃHcomb,H2
ᐃHcomb = ᐃHH − H +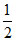 ᐃHO = O - 2ᐃHO − H - ᐃHvap = x1 +
ᐃHO = O - 2ᐃHO − H - ᐃHvap = x1 + 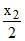 -2x3 - x4
-2x3 - x4
ᐃHcomb = ᐃHH − H +
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Boron exists in different allotropic forms. All allotropic form contains icosahedral units (icosahedral is a regular sha...The standard enthalpies of formation of NH3(g), CuO(s) and H2O(l) are −46, −155 and −285 kJ/mol, respectively. The entha...Standard molar enthalpy of formation of CO2 is equal to...The reaction of zinc metal with hydrochloric acid was used to produce 1.5 moles of hydrogen gas at 298 K and 1 atm press...In biological cells that have a plentiful supply of O2, glucose is oxidized completely to CO2 and H2O by a process calle...