ThermochemistryHard
Question
The enthalpy change (ᐃH) the reaction, N2(g) + 3H3(g) → 2NH3(g) is - 92.38 kJ at 398 K. The internal energy changed ᐃU at 298K is
Options
A.- 92.38 kJ
B.- 87.42kJ
C.- 97.34 kJ
D.- -89.9kJ
Solution
ᐃE = ᐃH - ᐃnRT
= - 92.38 -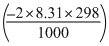
= - 92.38 = 4.95 = - 87.47kJ
= - 92.38 -
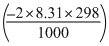
= - 92.38 = 4.95 = - 87.47kJ
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
From the following data, calculate the enthalpy change (in kJ/mol) for the combustion of cyclopropane(g) at 298 K. The e...Calculate ΔH for the following reaction at 298 K, Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2(g) from the following thermochemical...The standard enthalpy of formation of a substance...The molar heat capacities of A, B and C are in the ratio 1:2:3. The enthalpy change for the reaction A + B → C at temper...Heat of neutralisation of CsOH with all strong acid is 13.4 kcal mol-1. The heat released on neutralisation of CsOH with...