ThermochemistryHard
Question
How much energy is released when 6 moles of octane is burnt in air ? Given ᐃHfo for CO2(g), H2O(g) and C8H18(1) respectively are - 490, - 240 and + 160J/mol
Options
A.-6.2 kJ
B.- 37.4kJ
C.- 35.5kJ
D.- 20.0kJ
Solution
C + O2 = CO2 ᐃH = - 490 (i)
H2 +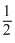 O2 = H2O ᐃH = - 240 (ii)
O2 = H2O ᐃH = - 240 (ii)
8C + 9H2 = C8H18 ᐃH = + 160 (iii)
2C8H18 + 25O2 = 16CO2 + 18H2O
The required reaction can be obtained by
2 × (iii) - 16(i) - 18(ii)
16C + 18H2 - 16C - 16O2 - 18H2 - 9O2
= 2C8H18 - 16CO2 - 18H2O
or - 25O2 = 2C8H18 - 16CO2 - 18H2O 2C8H18 + 25O2 = 16CO2 + 18H2O
[ᐃH = 2 × 160 - 16 × - 490 - 18 × - 240]
ᐃH = - 11840J = - 11.840 kJ for 2 moles of
Octance. Energy releced for 6 moles of
Octance. = - 11840 × 3 = - 35.5kJ
H2 +
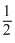 O2 = H2O ᐃH = - 240 (ii)
O2 = H2O ᐃH = - 240 (ii)8C + 9H2 = C8H18 ᐃH = + 160 (iii)
2C8H18 + 25O2 = 16CO2 + 18H2O
The required reaction can be obtained by
2 × (iii) - 16(i) - 18(ii)
16C + 18H2 - 16C - 16O2 - 18H2 - 9O2
= 2C8H18 - 16CO2 - 18H2O
or - 25O2 = 2C8H18 - 16CO2 - 18H2O 2C8H18 + 25O2 = 16CO2 + 18H2O
[ᐃH = 2 × 160 - 16 × - 490 - 18 × - 240]
ᐃH = - 11840J = - 11.840 kJ for 2 moles of
Octance. Energy releced for 6 moles of
Octance. = - 11840 × 3 = - 35.5kJ
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
At 300 K, the standard enthalpies of formation of C6H5COOH(s), CO2(g) and H2O(l) are −408, −393 and −286 kJ/mol, respect...ᐃHof of water is - 285.5 kJ mol-1. if enthalpy of neutralisation of monoacid strong base is - 57.3 kJ mol-1, Ȁ...The enthalpy change involved in the oxidation of glucose is −2880 kJ/mol. Twenty five per cent of this energy is availab...The enthalpy change when x g of phenol dissolves in y g of CHCl3 is given below at a certain temperature.x Y ΔH (kcal)0....If the enthalpy of formation and enthalpy of solution of HCl (g) are - 92.3 kJ/mol and - 75.14 kJ/mol respectively then ...