ThermochemistryHard
Question
The heat of neutralization of a strong base and a strong acid is 57 kJ. The heat released when 0.5 mole of HNO3 solution ia added to 0.20 moles of NaOH solution, is
Options
A.11.4 kJ
B.34.7 kJ
C.23.5 kJ
D.58.8 kJ
Solution
The chemical reaction between strong base and strong acid is a neutralisation reactionbetween H+ ion and OH- ion.
H+ + OH- → H2O + Heat
One mole of H+ and one mole OH- give 57 kJ.when 0.5 moles of H+ (from HNO3) reacts with 0.2 mole of OH- (From NaOH),0.2 mole of H+ is neutralised by 0.2 mole of OH- and 0.3 mole of H+ remains uncreated.
H+ + OH- → H2O
0.2mole 0.2mole
Heat evolved will be =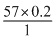 = 11.4kJ
= 11.4kJ
H+ + OH- → H2O + Heat
One mole of H+ and one mole OH- give 57 kJ.when 0.5 moles of H+ (from HNO3) reacts with 0.2 mole of OH- (From NaOH),0.2 mole of H+ is neutralised by 0.2 mole of OH- and 0.3 mole of H+ remains uncreated.
H+ + OH- → H2O
0.2mole 0.2mole
Heat evolved will be =
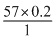 = 11.4kJ
= 11.4kJCreate a free account to view solution
View Solution FreeMore Thermochemistry Questions
When 1 g equivalent of strong acid reacts with strong base, the heat released is 13.5 kcal. When 1 g equivalent H2A is c...If the bond dissociation energies of XY, X2 and Y2 (all diatomic molecules) are in the ratio of 1:1:0.5 and ᐃfH fo...The enthalpy of formation of liquid methyl alcohol in kJ/mol, using the following data (in kJ/mol).Heat of vaporization ...Match the transformations in column I with appropriate options in column II...From the following data at 25o C, which of the following statement(s) is/are correct?½H2(g) + ½O2(g)→ OH(g): ΔHo = 42 kJ...