ThermochemistryHard
Question
If the bond dissociation energies of XY, X2 and Y2 (all diatomic molecules) are in the ratio of 1:1:0.5 and ᐃfH for the formation of XY is -200 kJ mole-1. The bond dissociation energy of X2 will be
Options
A.100 kJ mole-1
B.200 kJ mole-1
C.300 kJ mole-1
D.400 kJ mole-1
Solution
(None of the options is correct.)
XY → X(g) + Y(g) ; ᐃH = + a kJ /mole ........(i)
X22X ; ᐃH = + a kJ/mole ........(ii)
Y22Y ; ᐃH = + 0.5 a kJ/mole ........(ii)
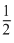 × (ii) +
× (ii) + 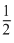 × (iii) - (i), Gives
× (iii) - (i), Gives
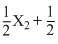 Y2 → XY ; ᐃH =
Y2 → XY ; ᐃH = 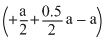 kJ / mole
kJ / mole
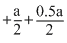 - a = - 200
- a = - 200
a = 800.
XY → X(g) + Y(g) ; ᐃH = + a kJ /mole ........(i)
X22X ; ᐃH = + a kJ/mole ........(ii)
Y22Y ; ᐃH = + 0.5 a kJ/mole ........(ii)
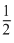 × (ii) +
× (ii) + 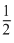 × (iii) - (i), Gives
× (iii) - (i), Gives 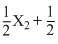 Y2 → XY ; ᐃH =
Y2 → XY ; ᐃH = 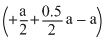 kJ / mole
kJ / mole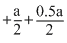 - a = - 200
- a = - 200 a = 800.
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
ᐃHfo(298K) of methanol is given by the chemical equation :...Reactions involving gold have been of particular interest to a chemist. Consider the following reactions.Au(OH)3 + 4HCl ...The enthalpies of formation of FeO(s) and Fe2O3(s) are −65.0 and −197.0 kcal/mol, respectively. A mixture of the two oxi...Calculate the enthalpy of formation (in kcal/mol) of anhydrous Al2Cl6 from the following data.2Al(s) + 6HCl(aq) → Al2Cl6...A solution was prepared by dissolving 7.45 g of KCl in 200 g of H2O in a calorimeter with a water equivalent of 25 g. Th...