ThermodynamicsHard
Question
Which of the following statement(s) is/are false ?
Options
A.All adiabatic processes are isoentropic (or isentropic) processes
B.When (ᐃGsystem) T,P < 0; the reaction must be exothermic
C.dG = VdP - SdT is applicable for closed system, both PV and non-PV work
D.the heat of vaporisation of water at 100oC is 40.6 kJ/mol. When 9 gm of water vapour condenses to liquid at 100oC of 1 atm, then ᐃSsystem = 54.42 J/K
Solution
(A) Reversible adiabatic process is isoentropic
(B) Reaction is spontaneous, need not be exothermic.
(C) Only when PV work is innvolved
(D) ᐃSsystem =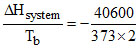 = - 54.42 J/K
= - 54.42 J/K
(B) Reaction is spontaneous, need not be exothermic.
(C) Only when PV work is innvolved
(D) ᐃSsystem =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In the reversible adiabatic expansion of an ideal monoatomic gas, the final volume is 8 times the initial volume. The ra...Which of the following sets does show the intensive properties?...An ideal gas ($\gamma$ = 1.5) undergoes a change in state such that the magnitude of heat absorbed by the gas is equal t...For the reaction of the one mole of zinc dust with one mole of sulphuric acid in a bombcalorimeter, ᐃU and w corre...An ideal gas undergoes isothermal expansion from (10 atm, 1 L) to (1 atm, 10 L) either by path–I (infinite stage expansi...