ThermochemistryHard
Question
Determine ᐃUo at 300 K for the following reaction using the listed enthalpies of reaction :
4CO(g) + 8H2(g) → 3CH4 (g) + CO2(g) + 2H2O(l)
C(graphite) + 1/ 2O2(g) → CO(g); ᐃH1o = - 110.5 kJ
CO(g) + 1/ 2O2(g) → CO2(g); ᐃH2o = - 282.9 kJ
H2(g) + 1/ 2O2(g) → H2O(l); ᐃH3o = -285.8 kJ
C (graphite) + 2H2(g) → CH4(g); ᐃH4o = -74.8 kJ
4CO(g) + 8H2(g) → 3CH4 (g) + CO2(g) + 2H2O(l)
C(graphite) + 1/ 2O2(g) → CO(g); ᐃH1o = - 110.5 kJ
CO(g) + 1/ 2O2(g) → CO2(g); ᐃH2o = - 282.9 kJ
H2(g) + 1/ 2O2(g) → H2O(l); ᐃH3o = -285.8 kJ
C (graphite) + 2H2(g) → CH4(g); ᐃH4o = -74.8 kJ
Options
A.-653.5 kJ
B.-686.2 kJ
C.-747.4 kJ
D.None of these
Solution
ᐃHo = - 3ᐃHo1 + ᐃHo2 + 2ᐃHo3 + 3ᐃHo4
= - 747.4 kJ
ᐃHo = ᐃUo + ᐃng RT; where ᐃng = - 8
- 747.4 = ᐃUo -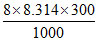
ᐃUo = - 727.44 kJ.
= - 747.4 kJ
ᐃHo = ᐃUo + ᐃng RT; where ᐃng = - 8
- 747.4 = ᐃUo -
ᐃUo = - 727.44 kJ.
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...Which of the following are true about resonance energy ?...A volume of 4.0 L of a mixture of ethane and methane gases on complete combustion at 300 K produced 6.0 L of carbon diox...From the following data, calculate the enthalpy change (in kJ/mol) for the combustion of cyclopropane(g) at 298 K. The e...Which of the following salts shall cause more cooling when one mole of the salt is dissolved in the same amount of water...