Chemical Kinetics and Nuclear ChemistryHard
Question
The rate constant of first order reaction is 3 × 10-6 per second. the initial concentration is 0.10 M.The intial rate is :
Options
A.3 × 10-7 moles/litre/sec
B.3 × 10-8 moles/litre/sec
C.3 × 10-5 moles/litre/sec
D.3 × 10-8 moles/litre/sec
Solution
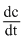 = k [c] × 3 × 10-6 × 0.1
= k [c] × 3 × 10-6 × 0.1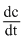 = × 3 × 10-7 mol litre-1sec-1
= × 3 × 10-7 mol litre-1sec-1Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Upon treatment with ammonical H2S , the metal ion that precipitates as a sulfide is -...Milk is colloid in which :...Bombardment of aluminium by a-particle leads to its artificial disintegration in two ways, (i) and (ii) as shown. Produc...Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrheni...Which of the following plots in correctly representing the variation in rate of vaporation and rate of condensation with...