JEE Main | 2014ThermodynamicsHard
Question
One mole of diatomic ideal gas undergoes a cyclic process ABC as shown in figure. The process BC is adiabatic. The temperatures at A, B and C are 400 K, 800 K and 600 K respectively. Choose the correct statement
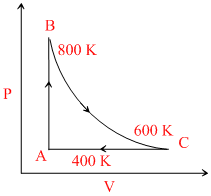

Options
A.The change in internal energy in whole cyclic process is 250R
B.The change in internal energy in the process CA is 700R
C.The change in internal energy in the process AB is - 350R
D.The change in internal energy in the process BC is - 500R
Solution
ᐃU = nCvᐃT = 1 × 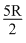 ᐃT
ᐃT
For BC, ᐃT = - 200 K
⇒ ᐃU = - 500R
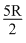 ᐃT
ᐃT For BC, ᐃT = - 200 K
⇒ ᐃU = - 500R
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A gas mixture contain 1 gm. H2 and 1 gm. He if temp. of gas mixture increase 0oC to 100o C at isobaric process. Than fin...A bimetallic strip is formed out of two identical strips - one of copper and the other of brass. The coefficients of lin...A quantity of heat requirad to change the unit mass of a solid substance to its liquid state, while the temperature rema...A carnot engine working between 300 K and 600 K has work out put of 800 J per cycle. The amount of heat and energy suppl...One mole of an ideal gas in initial state A undergoes a cyclic processABCA, as shown in the figure. Its pressure at A is...