Ionic EquilibriumHard
Question
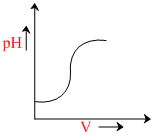
During titration of acetic acid with aq. NaOH solution, the neutralisation graph has avertical line. This line indicates

Options
A.alkaline nature of equivalence
B.acidic nature of equivalence
C.netural nature of equivalence
D.depends on experimental proceeding.
Solution
In the titration of weak acid (CH3COOH) with a strong base (NaOH), there is a change in the pH value at the end point. But it is not sharp due to weak ionisation of CH3COOH. Due to the excess of free base steep rise in pH which is indicated by vertical line. Hence the vertical line in the graph indicateds. alkaline nature of equivalence.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Calculate the degree of hydrolysis of 0.005 M-K2CrO4. For H2CrO4, Ka1 = infinite, Ka2 =$5 \times 10^{- 7}$....Which of the following statements about a weak acid strong base titration is/are correct ?...When 0.1 mol arsenic acids (H3AsO4) is dissolved in 1L buffer solution of pH = 8, which of the following hold good? For ...The solubility of Agl in Nal solutions is less than that in pure water because:...If pKb for fluoride ion at 25°C is 10.3, the ionization constant of hydrofluoric acid in water at this temperature is (l...