Ionic EquilibriumHard
Question
When 0.1 mol arsenic acids (H3AsO4) is dissolved in 1L buffer solution of pH = 8, which of the following hold good? For arsenic acid K1 = 2.5 × 10-4, K2 = 5 × 10-8, K3 = 2 × 10-13. [′<<′ sign denotes that the high concentration is at least more than 100 times the lower one]
Options
A.[H3AsO4] << [H2AsO4-]
B.[H2AsO4-] << [HAsO42-]
C.[HAsO42-] << [H2AsO4-]
D.[AsO43-] << [HAsO42-]
Solution
(A) 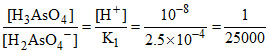 ∴ [H3AsO4] << [H3AsO4-].
∴ [H3AsO4] << [H3AsO4-].
(B)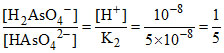
(C)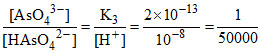 ∴ [AsO43-] << [HAsO42-].
∴ [AsO43-] << [HAsO42-].
(B)
(C)
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Graphite is :-...Select the correct statements : 1. pH of NaHCO3 solution can be given by 2. AI3+ ion is amphoteric3. KSP values of metal...Sr2+ forms a very unstable complex with NO3−. A solution that was 0.001 M-Sr(ClO4)2 and 0.05 M-KNO3 was found to have on...The correct order of acidic strength is :...An aqueous solution of a metal bromide MBr2 (0.04 M) is saturated with H2S. What is the minimum pH at which MS will prec...