ElectrochemistryHard
Question
Options
A.0
B.1.1
C.1.16
D.1.16
Solution
(1)
E = 1.1 -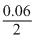 log
log 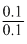
(2)
1.109 = 1.100 -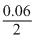 log
log 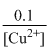
[Cu2+] = 0.2 M
áƒn = 0.2 × 0.1 - 0.1 × 0.1 = 0.01
w = 0.01 × 135 = 13.5 gm
(3)
ne =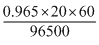 = 0.012
= 0.012
E = 1.1 -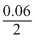 log
log 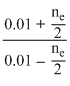
E = 1.1 -
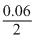 log
log 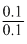
(2)
1.109 = 1.100 -
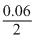 log
log 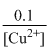
[Cu2+] = 0.2 M
áƒn = 0.2 × 0.1 - 0.1 × 0.1 = 0.01
w = 0.01 × 135 = 13.5 gm
(3)
ne =
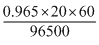 = 0.012
= 0.012E = 1.1 -
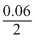 log
log 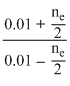
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard potential of the reaction H2O + e- → H2 + OH- at 298 K by using Kw (H2O) = 10-14, is :...For Zn2+ / Zn, Eo = - 0.76 V, for Ag+/Ag Eo = 0.799 V. The correct statement is -...ASelect the correct statement about product A....Standard electrode potentil data are useful for understanding the suitability of an oxidant in a redox titration. Some h...Expermentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+and M3+ in its oxide. Fracti...