Ionic EquilibriumHard
Question
The pH value of N/10 NaOH is :
Options
A.9
B.10
C.12
D.13
Solution
0.1 N NaOH ⇋ Na+ + OH-
Normality of NaOH = Molarity of NaOH =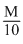
[OH-] = 10-1
[H+][OH-] = 10-1
[H+] =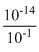 = 10-13
= 10-13
∴ pH = - log 10-13
∴ pH = 13
Normality of NaOH = Molarity of NaOH =
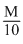
[OH-] = 10-1
[H+][OH-] = 10-1
[H+] =
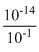 = 10-13
= 10-13∴ pH = - log 10-13
∴ pH = 13
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
If valence factor (n-factor) of compound NaHC2O4.2H2 C2O4 .3K2C2O4. 4Al2(C2O4)3.3FeC2O4 in acid base titration is x and ...The indicator constant for an acidic indicator, HIn is 5 × 10−6 M. This indicator appears only in the colour of acidic f...Consider an aqueous solution, 0.1 M each in HOCN, HCOOH, (COOH)2 and H3PO4, for HOCN, we can write Ka(HOCN) = . [H+] in ...Ka for HCN is 5 × 10-10 at 25oC. For maintaining a constant pH of 9, the volume of 5 M KCN solution required to be ...Four species are listed below i. HCO3- ii. H3O+ iii. HSO4- vi. HSO3F Which one of the following is the correct sequence ...