Mole ConceptHard
Question
Silver metal in ore is dissolved by potassium cyanide solution in the presence of air by the reaction
4 Ag + 8 KCN + O2 + 2H2O → 4 K[Ag (CN)2] + 4 KOH
4 Ag + 8 KCN + O2 + 2H2O → 4 K[Ag (CN)2] + 4 KOH
Options
A.The amount of KCN required to dissolve 100 g of pure Ag is 120 g.
B.The amount of oxygen used in this process is 0.742 g (for 100 gm pure Ag)
C.The amount of oxygen used in this process is 7.40 g (for 100 gm pure Ag)
D.The volume of oxygen used at STP is 5.20 litres.
Solution
(A), (C) and (D) Explanation :
4 Ag + 8 KCN + O2 + 2 H2O → 4 K[Ag(CN)2] + 4 KOH
⇒ 4 × 108 g of Ag reacts with 8 × 65 g of KCN
100 g of Ag reacts with
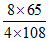 × 100 = 120
× 100 = 120
Hence, to dissolve 100 g of Ag , the amount of KCN required = 120 g
Hence, statement (A) is correct.
⇒ 4 × 108 g of Ag require 32 g of O2
1 g of Ag require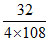 = 0.0740 g
= 0.0740 g
⇒ 100 g of Ag require = 7.4 g
Hence, choice (C) is correct.
Hence, volume of O2 required =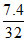 × 22.4 = 5.20 litre
× 22.4 = 5.20 litre
Hence, (A), (C), (D) are correct while (B) is incorrect.
4 Ag + 8 KCN + O2 + 2 H2O → 4 K[Ag(CN)2] + 4 KOH
⇒ 4 × 108 g of Ag reacts with 8 × 65 g of KCN
100 g of Ag reacts with
Hence, to dissolve 100 g of Ag , the amount of KCN required = 120 g
Hence, statement (A) is correct.
⇒ 4 × 108 g of Ag require 32 g of O2
1 g of Ag require
⇒ 100 g of Ag require = 7.4 g
Hence, choice (C) is correct.
Hence, volume of O2 required =
Hence, (A), (C), (D) are correct while (B) is incorrect.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
If we consider that , in place of ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mo...100 mL of 0.06 M Ca(NO3)2 is added to 50 mL of 0.06 M Na2C2O4. After the reaction is complete....In a compound C, H and N atoms are present in 9 : 1 : 35 by weight. Molecular weight of compound is 108. Molecular formu...A sample of ammonium phosphate, (NH4)3PO4, contains 6 moles of hydrogen atoms. The number of moles of oxygen atoms in th...10 mL of a gaseous hydrocarbon is exploded with 100 mL of oxygen. The residual gas on cooling is found tp measure 95 mL ...