Gaseous StateHard
Question
Two flasks of equal volume are connected by a narrow tube (of negligible volume) all at 27oC and contain 0.70 moles of H2 at 0.5 atm. One of the flask is then immersed into a bath kept at 127oC, while the other remains at 27oC. The number of moles of H2 in flask 1 and flask 2 are :
Options
A.Moles in flask 1 = 0.4, Moles in flask 2 = 0.3
B.Moles in flask 1 = 0.2, Moles in flask 2 = 0.3
C.Moles in flask 1 = 0.3, Moles in flask 2 = 0.2
D.Moles in flask 1 = 0.4, Moles in flask 2 = 0.2
Solution
Let vol. of each flask is ′V′ L, Initially
0.5 × 2V = 0.7 × 0.0821 × 300
V = 17.24 L
Let T is final temperature of flask when pressure in each flask becomes equal, it happens. when
nTotal = n1 + n2
 ⇒
⇒ 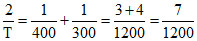
T =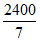 = 342.85 K
= 342.85 K
P × 2 × 17.24 = n1 × 0.0821 × 400 ⇒ n1 = 0.3
0.5714 × 17.24 = n2 × 0.0821 × 300 ⇒ n2 = 0.4
0.5 × 2V = 0.7 × 0.0821 × 300
V = 17.24 L
Let T is final temperature of flask when pressure in each flask becomes equal, it happens. when
nTotal = n1 + n2
T =
P × 2 × 17.24 = n1 × 0.0821 × 400 ⇒ n1 = 0.3
0.5714 × 17.24 = n2 × 0.0821 × 300 ⇒ n2 = 0.4
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Calculate the radius of He atoms if its Vander Waal′s constant ′b′ is 24 ml mol-1....Rate of diffusion of a gas is :...For the non-zero values of force of attraction between gas molecules, gas equation will be :...The order of ionisation potential between He+ ion and H-atom (both species are in gaseous state) is :-...Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...